In a corner of atmospheric science where dry-particles studies predominate, one ASR researcher just adds water

The sun radiates waves of electromagnetic energy to Earth through the vacuum of space. Only about half of that energy directly reaches land and ocean surfaces. Some is reflected or scattered back into space.
But about 20 percent of that solar radiation is absorbed by clouds and the atmosphere. This fraction is what captivates Chris Cappa, professor and department chair in civil and environmental engineering at the University of California, Davis.
For a decade or more, one issue of absorption, in particular, has informed Cappa’s work and that of his research group: How particles within the atmosphere are mixed and internally organized in ways that affect their ability to absorb or scatter sunlight.
“What really matters is how much sunlight makes it to the surface, or goes back into space, or is absorbed by the atmosphere itself,” he says.
It is important to understand this process, since the more solar radiation particles absorb, the more the atmosphere warms. Take, for instance, black and brown carbon, which absorb sunlight strongly and are abundantly emitted during wildfire and other biomass burning events. (By contrast, purely scattering particles have a cooling effect.)
In turn, absorption and related atmospheric warming impact atmospheric dynamics, including storm-making convective processes.
Studying absorption, of course, is part of investigating atmospheric aerosols. These ultrafine liquid and solid particles influence cloud formation, precipitation, and the Earth’s surface energy budget.
Such particles can be less than a micron wide. Thus, the mighty small have a mighty effect on the planet.
Dynamic, Liquid, Hydrophilic
Lately, Cappa has drilled down into one aspect of particle mixing and structure: the role that water plays in how much sunlight a particle absorbs.
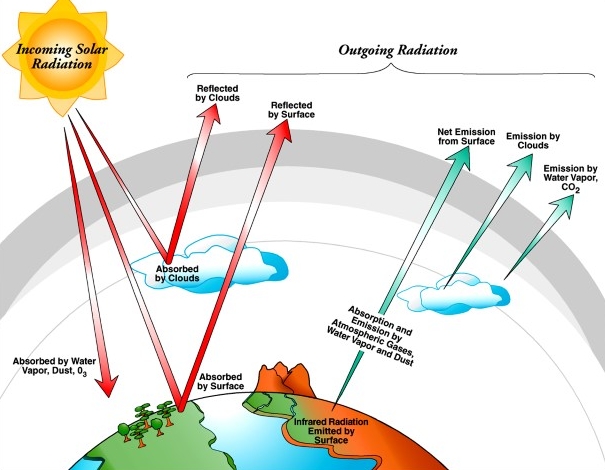
“The vast majority of studies focus on dry particles and how they absorb energy at different wavelengths,” he says. (Most of the sun’s radiant energy arrives on Earth in the visible or near-infrared spectra of light, and less than 10 percent as ultraviolet light.)
Studies of dry particles are essential and provide valuable insights into particles’ general behavior during sunlight absorption, says Cappa. “But water is just fundamentally different than these (dry) components: dynamic, liquid, and hydrophilic. It may take up a large volume of the particle.”
When water mixes with black and brown carbon, for instance, “that might cause everything to be more fluid and change the morphology (shape) of the particles,” he says.
So far, scientists have a limited experimental understanding of the role water plays in how particles absorb sunlight.
To further investigate the role of water in this process, Cappa and others have two projects underway that are funded by the Atmospheric System Research (ASR) program at the U.S. Department of Energy (DOE). The work also uses data from round-the-clock observatories run by Atmospheric Radiation Measurement (ARM), a DOE user facility.
Both of the ASR projects target how particles either absorb or scatter sunlight, a parameter used in models that remains “one of the most uncertain … in understanding climate change,” says Cappa.
The Power of Humidity
The two ASR projects have a common aim: investigate how the interaction between aerosols and radiation depends on relative humidity.
The first, “Characterizing the Impact of Water Uptake on Light Absorption by Aerosol Particles” (2019-2022), does so in a laboratory setting.
The second, “Characterization of Carbonaceous Aerosols during TRACER-CAT” (2020―2023), which just started, will bring these inquiries into the field as part of the Tracking Aerosol Convection Interactions Experiment (TRACER), a year-long, all-season ARM campaign scheduled to begin on October 1, 2021, in Houston, Texas.

Getting into the field is a step forward, says Cappa, and will supplement a lab approach to developing “the process-level understanding necessary to improving models.”
The second project also involves a strong collaboration with Los Alamos National Laboratory in New Mexico and with the lab’s Allison Aiken, an aerosols research scientist.
Both current ASR projects have their genesis in a third, earlier ASR project led by Cappa, “Characterizing and Understanding Aerosol Optical Properties: CARES.” This 2012―2015 study looked at the scattering and absorbing properties of atmospheric aerosol properties and their direct effect on solar radiation. (The “indirect effects” of such particles, also investigated, relate to how such particles influence clouds.)
Importantly, this foundational ASR project took into account hygroscopicity, the tendency for a solid to take up water from its environment.
Cappa and his co-researchers drew the bulk of their data for the 2012―2015 ASR project from another ARM field campaign, the 2010 Carbonaceous Aerosols and Radiative Effects Study (CARES). They produced a 2015 study that explicitly looked at hygroscopicity measurements and a summary paper in 2016.
A co-author with Cappa on both papers was UC Davis environmental toxicologist Qi Zhang. She has remained his co-principal ASR investigator as they study the effect of water uptake on the scattering or absorption properties of aerosols.
A New Instrument Combination
Bringing water to absorption studies presents a measurement challenge because it requires adding high humidity to dry-measurement protocols.
Cappa, Zhang, and others have addressed that hurdle by taking two familiar technologies “and bringing them together in one package” and adding the capacity to measure temperature and humidity, says Cappa.
One of the instruments employs Cavity Attenuated Phase Shift (CAPS) spectroscopy to measure aerosol light extinction. Both particle absorption and scattering contribute to the extinction (disappearance) light in the atmosphere. In the case of visible light, optical extinction (the total attenuation of light) is the sum of absorption and scattering together.
Cappa and the others worked with Timothy Onasch and Andrew Freedman at Aerodyne Research, Inc. in Massachusetts to combine CAPS technology with a Single Scattering Albedo (SSA) device that distinguishes between scattering and absorption.
The resulting CAPS-SSA device is already widely used in surface and aerial observations to measure dry particles. With the help of Aerodyne, the ASR researchers have modified it to add temperature and humidity controls.
It’s all in service of their primary aim: to see how water affects absorption in particles.
Improving Models

During TRACER, the plan is to deploy the new hybrid instrument along with a standard suite of devices that measure light absorption, light extinction, black carbon concentrations, and the composition of light-absorbing particles.
But the big new hope is to be able to measure the effects of water―particularly how much water increases absorption.
About that, “there is a dearth of experimental information,” says Cappa. “It has really limited our ability to test and refine models.”
In laboratory work to date, Cappa and the others have done experiments with various black carbon and brown carbon surrogates, which take up water to different degrees.
“So far,” he says, “we observe that when we humidify, we see notable increases in absorption. Water uptake appears to be an important contributor to absorption by ambient particles.”
Still, adds Cappa, “we lack a process-level understanding of what drives that―so we can have greater confidence in what our (global warming) predictions are.”
On the modeling side, with a focus on black and brown carbon, the Cappa ASR team has tied its results to simple theoretical frameworks to test modeled systems and reproducibility.
“The goal is to improve the predictive acuity of a model,” says Cappa, echoing one ASR mission, “and to provide parameterizations within a model that are more physically realistic and tied to better observations.”
Finding Atmospheric Chemistry

A native of suburban Chicago and the son of an engineer and an accountant, Cappa enrolled at Hope College in Michigan (B.S., 2000) with an eye on pre-med and a biology track. He was a first-semester freshman when he sat in on a seminar-series lecture by atmospheric chemist Matt Elrod, now at Oberlin College and Conservatory in Ohio.
The talk was enough to steer Cappa towards chemistry (his major) and environmental science (his minor).
“It was then I knew I wanted to do atmospheric chemistry, he remembers, and it seemed “relevant and important.”
Elrod was in his first year at Hope, too, as a teacher, and looking to fill out a research group. Cappa joined.
“A fantastic thing about Hope was that lots of (undergraduates) were doing very high-quality research, especially in chemistry,” he says.
Later, as a doctoral student in chemistry at the University of California, Berkeley (PhD, 2005), Cappa gravitated to physical chemistry, the orbit of two of his most influential professors, Ronald Cohen and Richard Saykally.
He was also influenced from afar by the idea that so many prominent scientists started as physical chemists, including the late Mario Molina at the Massachusetts Institute of Technology―a Nobel laureate for his ozone-layer depletion work.
“It was the ability to blend physical chemistry with the world of atmospheric chemistry was really enticing to me,” says Cappa, whose doctoral dissertation in part was on the physical chemistry that governs the isotopic fractionation of water during evaporation.
As a postdoc at the University of Colorado Cooperative Institute for Research in Environmental Sciences and NOAA (2005―2007), Cappa did work on the optical properties of aerosols with mentor A.R. “Ravi” Ravishankara.
The experience, he says, “really put me back into the world of atmospheric chemistry.”
Exhaled Particles

In his very full research life outside of ASR projects, atmospheric chemistry has led Cappa to some diverse explorations, including work not long ago that reported on the surprisingly large impact that consumer products (including paint and perfume) have on urban air quality.
He also studies the junction of air quality and climate, a theme that has run through his research for years. Just before CARES, for instance, Cappa took part in California Nexus (CalNex 2010), an air-and-sea NOAA-National Science Foundation field campaign.
In the last few years, he has also done measurement work on respiratory aerosols.
Even before the pandemic, Cappa and colleagues at UC Davis and the Icahn School of Medicine at Mount Sinai in New York studied the generation and exhalation of respiratory aerosol particles and the relative effectiveness of various mask types. This emphasis on outward aerosol emissions, as in one recent paper, is an unusual other-directed approach, says Cappa, “versus the usual approach of protecting the wearer.”
He and colleagues explore questions such as: What factors make someone a COVID-19 super-spreader? Does the language you speak matter in measured aerosols output? (Not as much as loudness, it turns out.) How can researchers overcome the challenge of measuring the efficacy of cloth masks, which shed fibers as they emit aerosols? And how about a mask for singers? (In January 2021, Cappa started aerosol-emissions testing on one design with singers from the San Francisco Opera.)
A glimpse at Cappa’s Twitter account (@CappaSnappa) reveals a few interests (pollution, climate, cats). But you have to look harder for mentions of other pastimes, including acoustic guitar and running half-marathons. (“I used to be fast,” he says.)
Then comes a Cappa statement that, one suspects, will not survive scientific scrutiny.
“I’m a dilettante who is marginally OK at many things,” he says, “but not very good at anything.”
# # #Author: Corydon Ireland, Staff Writer, Pacific Northwest National Laboratory
This work was supported by the U.S. Department of Energy’s Office of Science, through the Biological and Environmental Research program as part of the Atmospheric System Research program.

